A somatization comorbidity phenotype impacts response to therapy
Por um escritor misterioso
Last updated 06 novembro 2024
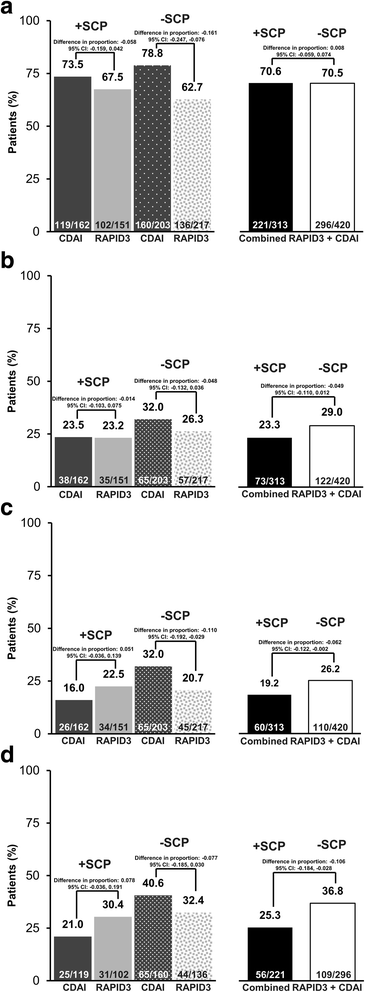
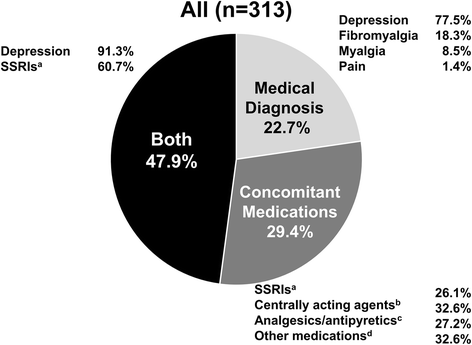
Background Comorbidities may contribute to disease activity and treatment response in rheumatoid arthritis (RA) patients. We defined a somatization comorbidity phenotype (SCP) and examined its influence on response to certolizumab pegol (CZP) using data from the PREDICT trial. Methods Patients in PREDICT were randomized to the patient-reported Routine Assessment of Patient Index Data 3 (RAPID3) or physician-based Clinical Disease Activity Index (CDAI) for treatment response assessment. Post-hoc analyses identified patients with the SCP, which included diagnosis of depression, fibromyalgia/myalgias, and/or use of medications indicated for treatment of depression, anxiety, or neuropathic pain. The effect of the SCP on RAPID3 or CDAI response at week 12 and low disease activity (LDA; Disease Activity Score in 28 joints based on erythrocyte sedimentation rate ≤ 3.2) at week 52, in week-12 responders, was analyzed using non-parametric analysis of covariance (ANCOVA). Results At baseline, 43% (313/733) of patients met the SCP classification. Patients with the SCP were 9% more likely to withdraw from the trial. American College of Rheumatology 20% (ACR20), ACR50, and ACR70 responses were 5–14% lower among those with the SCP, and 11% more patients reported adverse events (AEs). Patients without SCP in the CDAI arm were twice as likely to achieve LDA at week 52 compared with those with SCP (32% versus 16%). No differentiation by SCP was observed in the RAPID3 arm (pooled result 21.5%). Conclusions We operationalized a potentially important somatization comorbidity phenotype in a trial setting that was associated with a substantially lower likelihood of treatment response and a higher frequency of AEs. Including large numbers of patients with this phenotype in RA trials may reduce the measured clinical effectiveness of a new molecule. Trial registration ClinicalTrials.gov, NCT01255761 . Registered on 6 December 2010.
Biomedicines, Free Full-Text

Preliminary proposal for a revision to the Diagnostic and Statistical
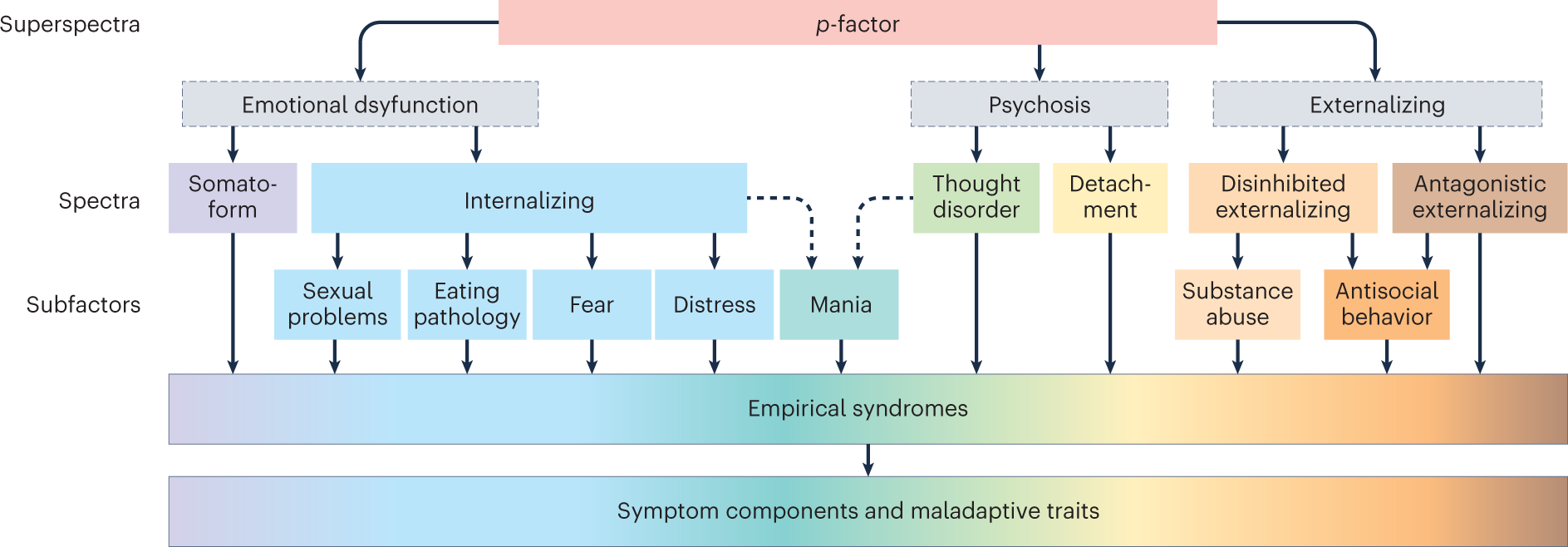
Precision behavioral phenotyping as a strategy for uncovering the biological correlates of psychopathology

Towards better care for women with schizophrenia-spectrum disorders - The Lancet Psychiatry
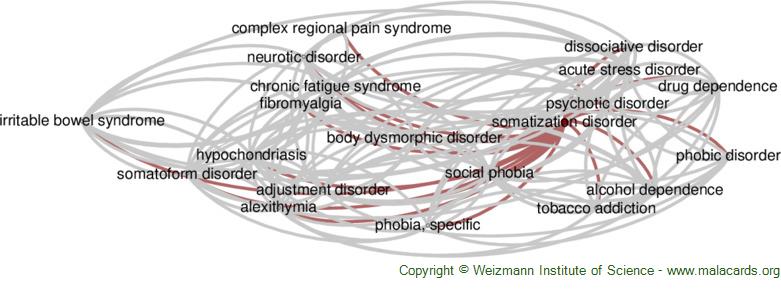
Somatization Disorder disease: Malacards - Research Articles, Drugs, Genes, Clinical Trials

A model of phenotype relationship among several disorders frequently
Nutrients, Free Full-Text

Functional gastrointestinal disorders: advances in understanding and management - The Lancet

Long COVID: pathophysiological factors and abnormalities of coagulation: Trends in Endocrinology & Metabolism
IJMS, Free Full-Text

Healthcare, Free Full-Text
A somatization comorbidity phenotype impacts response to therapy in rheumatoid arthritis: post-hoc results from the certolizumab pegol phase 4 PREDICT trial, Arthritis Research & Therapy
Nutrients, Free Full-Text
Recomendado para você
-
 SCP-076-2, Villains Wiki06 novembro 2024
SCP-076-2, Villains Wiki06 novembro 2024 -
 SCP-076 ABLE (SCP Animation)06 novembro 2024
SCP-076 ABLE (SCP Animation)06 novembro 2024 -
 JSCAPE MFT Server06 novembro 2024
JSCAPE MFT Server06 novembro 2024 -
 Day 76: Jar Creature 1 Rigged and Animated - Alfred Reinold Baudisch06 novembro 2024
Day 76: Jar Creature 1 Rigged and Animated - Alfred Reinold Baudisch06 novembro 2024 -
 TSXSCPCD1030 - Telemecanique - Tsx Scp CD 1030 / Modicon Cable Linkage 9 10/12ft06 novembro 2024
TSXSCPCD1030 - Telemecanique - Tsx Scp CD 1030 / Modicon Cable Linkage 9 10/12ft06 novembro 2024 -
 эпичиские мини игры - KoGaMa - Play, Create And Share Multiplayer06 novembro 2024
эпичиские мини игры - KoGaMa - Play, Create And Share Multiplayer06 novembro 2024 -
 Shin Megami Tensei: Devil Summoner 2 Part #16 - Kasumidai reached06 novembro 2024
Shin Megami Tensei: Devil Summoner 2 Part #16 - Kasumidai reached06 novembro 2024 -
Paintings — Bailie Studios06 novembro 2024
-
 List of Authors - Escape from Site 1906 novembro 2024
List of Authors - Escape from Site 1906 novembro 2024 -
 Buy SAYAN CREATION Color Block Bollywood Pure Cotton Multicolor06 novembro 2024
Buy SAYAN CREATION Color Block Bollywood Pure Cotton Multicolor06 novembro 2024
você pode gostar
-
 Sub-19: Portugal volta a vencer na Nations Cup06 novembro 2024
Sub-19: Portugal volta a vencer na Nations Cup06 novembro 2024 -
 Já é Natal: as atrações natalinas nos shoppings cariocas06 novembro 2024
Já é Natal: as atrações natalinas nos shoppings cariocas06 novembro 2024 -
 IRON FIST - MARVELS IRON FIST SEASON 1 - COMPLETE FIRST SERIES **BRAND NEW DVD06 novembro 2024
IRON FIST - MARVELS IRON FIST SEASON 1 - COMPLETE FIRST SERIES **BRAND NEW DVD06 novembro 2024 -
 Conta de Roblox com mais de 100.000 robux - Videogames - Capão06 novembro 2024
Conta de Roblox com mais de 100.000 robux - Videogames - Capão06 novembro 2024 -
 Crítica Super Mario Bros Filme evoca tudo aquilo que torna Mario06 novembro 2024
Crítica Super Mario Bros Filme evoca tudo aquilo que torna Mario06 novembro 2024 -
 That one sonic colors render : r/SonicTheHedgehog06 novembro 2024
That one sonic colors render : r/SonicTheHedgehog06 novembro 2024 -
 Lung Cancer Awareness Month 202306 novembro 2024
Lung Cancer Awareness Month 202306 novembro 2024 -
 Resident Evil: 9 Things Infinite Darkness Changes About Claire06 novembro 2024
Resident Evil: 9 Things Infinite Darkness Changes About Claire06 novembro 2024 -
 Bola Pokémon Verde Sunny Ataque Surpresa Toxel - Pequenos Travessos06 novembro 2024
Bola Pokémon Verde Sunny Ataque Surpresa Toxel - Pequenos Travessos06 novembro 2024 -
 IGI 2 PC Game Free Download - IPC Games06 novembro 2024
IGI 2 PC Game Free Download - IPC Games06 novembro 2024
